|
4/21/2024 0 Comments Periodic tabl with molar mass 
nist_periodictable_july2019P E R I O D I C T A B L E Atomic Properties of the Elements.P E R I O D I C T A B L E Atomic Properties of the Elements Use the atomic mass of sulfur from the periodic table and the following atomic masses to determine whether these data are accurate assuming that these are the only isotopes of sulfur 31. Using a mass spectrometer a scientist determined the percent abundances of the isotopes of sulfur to be 95.27% for 32 S 0.51% for 33 S and 4.22% for 34 S.Howard_Universit圜hapter 1.6 Isotopes and Atomic Masses - Chemistry LibreTexts.Views 968.5KĬhapter 1.6 Isotopes and Atomic Masses - Chemistry LibreTexts This number is usually expressed as a decimal rather than as a whole. This is almost always written as a number at the bottom of the elements square on the table under its one or two letter chemical symbol. Most standard periodic tables list the relative atomic masses (atomic weights) of each element. Locate atomic mass on the periodic table.Calculate-Atomic-Mass3 Ways to Calculate Atomic Mass - wikiHow.ģ Ways to Calculate Atomic Mass - wikiHow damAmerican Chemical Society 57-71 Lithium 6.94 Beryllium 9.012 Sodium 22.99 Magnesium 24.31 Francium (223) Radium (226) Rutherfordium (265) Dubnium.damACS Periodic Table of Elements (PDF). 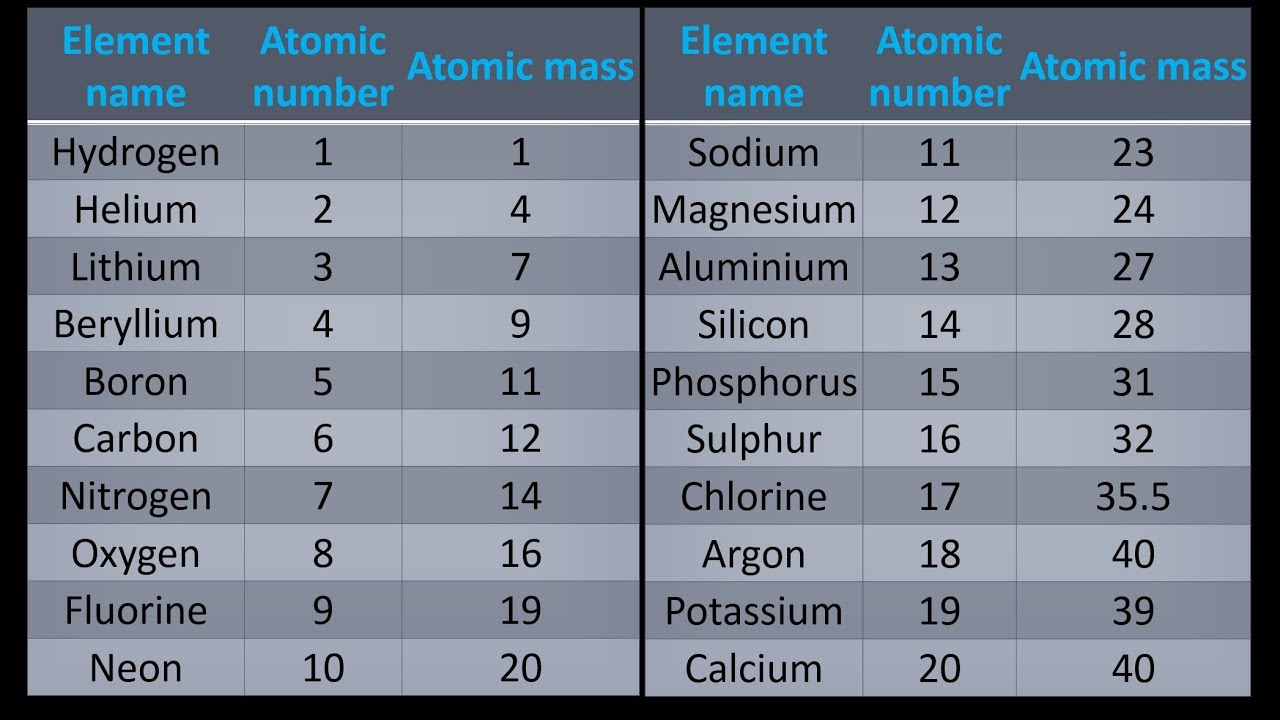
Where more than one isotope exists the value given is the abundance weighted average. This is approximately the sum of the number of protons and neutrons in the nucleus. Relative atomic mass The mass of an atom relative to that of carbon-12. element CachedDensity is the mass of a substance that would fill 1 cm 3 at room temperature.elementGold - Element information properties and uses | Periodic Table.Periodic table with molar mass Gold - Element information properties and uses | Periodic Table 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
